Mitral regurgitation (MR) is the primary cause of moderate-to-severe valvular disease in patients older than 55 years in the US.1 MR occurs when valve distortion leads to a retrograde blood flow from the left ventricle to the left atrium during systole. Proper functioning of the mitral valve requires an appropriate anatomical and physiological relationship between the mitral annulus, anterior and posterior leaflets, chordae tendineae, left atrium, and left ventricle.
MR can be classified as organic (primary) or functional (secondary). Organic MR can occur due to multiple causes, including degenerative valve disease, myxomatous disease, chordal rupture and infective endocarditis. MI and ischaemic heart disease is a common cause of secondary MR, and is associated with a higher rate of cardiac-related morbidity and mortality than in patients with primary mitral valve disease.2
Ischaemic MR can occur in both an acute and chronic fashion. When MR occurs in the acute setting, it can be associated with papillary muscle rupture secondary to MI. These patients can present with cardiogenic shock and congestive heart failure, as the body does not have time to compensate for the changes in haemodynamics. Treatment is aimed at the management of shock, and these patients often require emergent surgical intervention. Acute MR can also manifest with more subtle changes, such as annular dilatation and subvalvular apparatus dysfunction.
Revascularisation, whether surgical or percutaneous, should be accomplished to limit the extent of infarction. Consideration should also be given to performing a concomitant mitral valve repair or replacement for moderate-to-severe and severe MR in the acute setting. While transcatheter edge-to-edge repair (TEER) has been described in limited case reports of acute ischaemic MR, the role for this is not well established. In contrast, chronic MR is often the result of a long-standing process in which left ventricular remodelling has occurred, leading to papillary muscle displacement. The result is annular dilatation with chordal restriction of leaflet motion, leading to incomplete mitral valve coaptation. Clinically, these patients can have a compensated process with preserved cardiac output or a decompensated process with reduced cardiac output. This article focuses on functional MR as it relates to chronic ischaemic cardiomyopathy and the treatment options available.
Diagnosis
Diagnosis should begin with a patient medical history, complete physical examination and laboratory studies. The classic clinical finding on physical examination is a holosystolic murmur radiating to the left axilla. Given that the severity of the regurgitant jet can vary under different loading conditions, an increase in the murmur can be elucidated utilising manoeuvres that increase left ventricular afterload. Similarly, a decrease in afterload may decrease the regurgitant volume. Symptoms and clinical signs of heart failure can be present if the patient is not well compensated. Additionally, the heart rhythm, rate and presence of a previous MI can affect the degree of MR. ECG changes usually indicate the presence of an old infarct, and AF is common. Chest X-ray can show an enlarged heart if there is severe left ventricular enlargement.
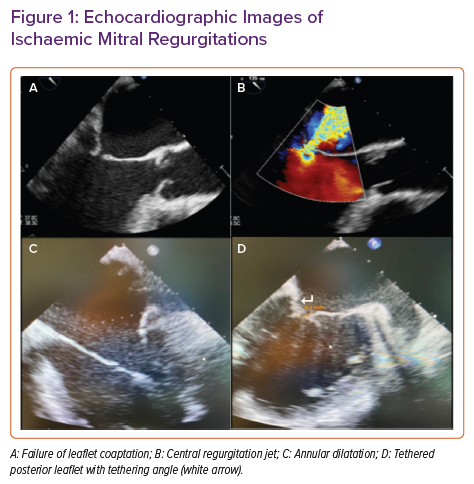
Transthoracic echocardiogram is the gold standard often initially used to characterise the directionality, eccentricity and severity of the regurgitant flow. Additionally, it can provide structural information regarding leaflet coaptation and tethering, and define the mechanism of MR (Figure 1). It allows for the assessment of left ventricular function and the determination of regional wall motion abnormalities.
Cardiac MRI can be adjunctive in the work-up of mitral valve disease, including the quantification of MR. Severe MR is defined by a regurgitant volume >60 ml, regurgitant fraction >50%, effective regurgitant orifice >0.4 cm2, jet area >40% of the left atrial area or vena contracta width >0.7 cm.3,4 Ejection fraction (EF) is often a misrepresentation of cardiac functioning in these patients due to increased preload in the left atrium.
Transoesophageal echocardiogram (TEE) is a third modality useful in the diagnosis of ischaemic MR and can help further identify the role of certain therapeutic options. The use of general anaesthesia in the operating room may decrease afterload, and this should be taken into consideration if a variation is noted from the preoperative transthoracic echocardiogram and intraoperative TEE.
Finally, coronary angiography is also an important diagnostic tool to assess for ongoing coronary disease that may be amenable to intervention.
Medical Management
Once the diagnosis is made, the severity of MR needs to be established. Patients who are well compensated without heart failure and with mild and moderate MR do not require invasive therapy. However, the management of patients who present with heart failure and moderate-to-severe MR has evolved over time.
The first-line approach to treatment in patients with heart failure and ischaemic MR is guideline-directed medical therapy (GDMT). Treatment, managed by a heart failure team, with β-blockers, angiotensin-converting enzyme inhibitors, diuretics, sodium-glucose cotransporter 2 inhibitors and angiotensin receptor blockers (with or without a neprilysin inhibitor) has been shown to improve survival.4,5 Underlying coronary artery disease should be addressed with optimal medical management and lifestyle changes to prevent ischaemia and resultant myocardial remodelling. AF can increase left atrial preload, leading to atrial dilatation and subsequent increased severity of MR. Anti-arrhythmic medications and cardiac resynchronisation should be pursued, as restoring normal sinus rhythm decreases the severity of MR.6 In the event that maximal medical therapy is achieved with ongoing symptoms and residual MR, additional surgical and/or transcatheter interventions can be considered.
Operative Intervention
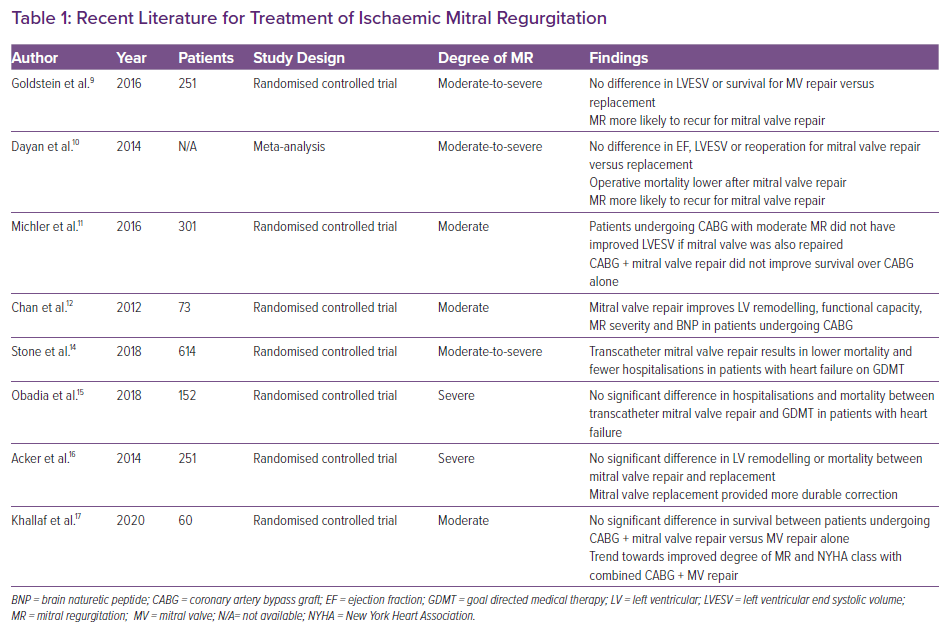
The recommendation for surgical intervention for the treatment of MR varies based on patient characteristics, as well as the underlying pathophysiology and presentation of valvular disease (Table 1). Surgical intervention for severe primary MR is often recommended regardless of EF.7 Intervention for secondary MR, however, is reserved for patients with persistent symptoms despite GDMT who have moderate-to-severe or severe MR. A 3-month trial of GDMT should be pursued prior to consideration of operative intervention.7
The pathology of ischaemic MR is largely due to increases in annular dimensions with resultant lack of leaflet coaptation. As a result, the central MR jet will often confirm a failure of leaflet coaptation based on annular size, rather than organic valvular dysfunction. In patients with minor left ventricular and subvalvular apparatus remodelling, and those in which gross inspection of the leaflets are normal, consideration for mitral valve repair should be given. This is accomplished with a downsized annuloplasty ring to reduce the annular dilatation and improve leaflet coaptation.
In contrast, mitral valve repair is less favourable in patients with posterolateral wall dysfunction due to prior infarction, and advanced leaflet tethering and papillary muscle displacement where adequate coaptation is less likely to be durable with an annuloplasty ring alone. Chordal-sparing mitral valve replacement is preferred over mitral valve repair in these patients, given the high reoperation rates seen with mitral valve repair failures. In these cases, strong consideration should be made for replacement of the valve upfront, even when the leaflets appear normal, given the degree of leaflet tethering and ventricular remodelling.
The American Heart Association/American College of Cardiology guidelines published in 2020 recommend operative intervention for patients with chronic severe symptomatic secondary MR with atrial annular dilatation and preserved EF.7,8 Consideration of concomitant procedures, such as ligation of the left atrial appendage, radiofrequency ablation or maze procedure, should be undertaken when performing a mitral valve operation on patients with associated arrhythmias.
There have been numerous studies examination the benefit of repair versus replacement in ischaemic MR. One study comparing mitral valve repair with mitral valve replacement in patients with severe ischaemic MR showed decreased rates of moderate-to-severe MR after surgery, fewer adverse events and less frequent postoperative readmissions for patients who underwent mitral valve replacement.9 Conversely, a meta-analysis from 2014 reported that patients with ischaemic MR undergoing mitral valve repair showed similar outcomes between the two groups. Patients undergoing operative mitral valve repair had lower operative mortality, but higher long-term recurrence rates of mitral regurgitation. When repairs performed before 1998 were excluded, the operative mortality was found to be the same for the two techniques. No differences in postoperative change in EF, ventricular dimensions, New York Heart Association class or reoperation rates were found between the two groups.10
Patients with MR and coronary artery disease with viable myocardium and suitable targets are often additionally treated with coronary artery bypass grafting. As patients with severe secondary MR and coronary artery disease are recommended to have a valvular operation in conjunction with bypass surgery, it is essential to ensure evaluation with left heart catheterisation in the preoperative assessment.7 In patients with moderate ischaemic MR, some studies have shown that addressing the myocardial ischaemia with isolated coronary artery bypass grafting and deferring mitral valve intervention has no negative effect on survival.11 This technique is used in this particular patient population with the anticipation of improvement of the MR once myocardial ischaemia is reversed and cardiac remodelling can occur, which will often lead to improvement of the mitral regurgitation.
Controversy surrounding this technique exists due to conflicting findings in the literature related to this patient population. One study included 73 patients with moderate ischaemic MR and EF >30% who were randomised to receive either a revascularisation procedure alone or coronary artery bypass grafting with concomitant mitral valve repair. Patients who underwent coronary artery bypass grafting and MV repair were shown to have greater peak oxygen consumption, and greater improvements in the left ventricular end systolic volume index, regurgitant volume and B-type natriuretic peptide levels after 1 year. Notably, survival was similar in the two groups.12 As such, patients with asymptomatic, moderate MR undergoing cardiotomy for alternative indications, such as coronary artery bypass grafting or aortic valve replacement, may be considered for intervention on the mitral valve at the time of operation based on the risk associated with the patient.
Catheter-based Treatment
Endovascular techniques for the management of MR have advanced over the last decade, and previously were pursued in patients with symptomatic severe MR who had a prohibitive operative risk secondary to medical comorbidities.7,8 During these procedures, venous access followed by a transseptal puncture is achieved, and a transcatheter edge-to-edge repair is performed using a device that reapproximates the central portion of the anterior and posterior mitral valve leaflets. The resultant anatomical configuration is similar to that of an operative Alfieri stitch.13
A 2018 study evaluated outcomes in 614 patients with heart failure and ischaemic MR randomly assigned to intervention with TEER in addition to GDMT versus GDMT alone. The results of this study, the COAPT Trial, showed improved survival at 2 years in the group who underwent TEER as opposed to GDMT alone.14 Prior to intervention, patients must be carefully assessed for candidacy. TEE should be performed to assess anatomical variables, including leaflet length, orifice area, presence of calcium burden and left ventricular cavity size, as these may be prohibitive to TEER.8 In addition, a coronary angiography should be considered to rule out underlying ischaemia that may be treatable and contributing to MR.
Similar to trends in other transcatheter-based valvular procedures, such as transcatheter aortic valve replacement, TEER has increased in utilisation over the past several years to include some patients outside those originally considered medically prohibitive to operative intervention. Some conflicting data have emerged, as published in the MITRA-FR trial, which should be mentioned.15 In this large-volume randomised control trial, there was no improvement in hospital admissions or death after 1 year among patients with severe secondary MR treated with percutaneous mitral valve repair plus GDMT versus medical therapy alone.
Despite these data, the candidacy for TEER has been expanded. In accordance with the most recent American Heart Association/American College of Cardiology guidelines in 2020, patients with chronic secondary MR and persistent symptoms despite GDMT with left ventricular EF of 20–50%, left ventricular end-systolic diameter <70 mm or pulmonary artery systolic pressure <70 mmHg can be treated with TEER.7 In addition, patients must be evaluated at a centre with a multidisciplinary team with expertise in heart failure and mitral valve disease.
Given the success of TEER in the treatment of patients with severe functional MR, numerous new devices are being investigated for improved device delivery and reduced damage to the native leaflets. As more advanced devices become available, the indication for these therapies is likely to increase even further. In those patients who prove to be high or extreme risk for surgery and otherwise not candidates for TEER due to inadequate leaflet length, calcium burden or other prohibitive reasons, transcatheter mitral valve replacement is being studied as an alternative therapy. The results of this are yet to be determined and require further investigation.
Conclusion
Proper functioning of the mitral valve requires a complex interplay between all involved structures. Multiple therapies are available to alleviate MR, including medical management, TEER, mitral valve repair and mitral valve replacement. Treatment should be undertaken after a thorough understanding of the underlying disease process leading to MR is obtained. Medical management with goal-directed therapy should be utilised in patients with heart failure and mild-to-moderate regurgitation. Transcatheter approaches are typically used in patients with prohibitive operative risk, although their use is expanding, especially in those with functional MR who are not responding to GDMT. Ischaemic MR is a complex process with debate in the surgical literature as to the optimal treatment pathway. It is generally accepted that patients with mild-to-moderate disease can avoid valve intervention if successful revascularisation is performed. A higher consideration should be given to valve replacement over repair in patients with severe MR in the setting of myocardial ischaemia. Operative course must be personalised to each patient, and continues to develop with improving technologies and ongoing research on optimal treatment.