Since its inception, cardiovascular critical care has witnessed an increase in the complexity of its patient population and the therapies available. The initiation and management of anticoagulation is a fundamental practice for a wide variety of indications, including the management of patients with acute MI, stroke prevention in patients with AF or mechanical valves, as well as the prevention of device thrombosis and thromboembolic events with the use of mechanical circulatory support and ventricular assist devices.
Management of peri- and postoperative cardiovascular patients also mandates an ability to adequately assess for and to pivot between optimal haemostatic conditions to anticoagulation states suitable to mitigate blood loss. The frequent use of antiplatelet and anticoagulation therapy, in addition to the presence of concomitant conditions that may lead to a propensity to bleed, such as renal and liver dysfunction, present unique challenges requiring a heavy reliance on testing that allows the cardiac intensivist to strike a delicate balance to avoid thrombotic and bleeding events.1
Common (or conventional) coagulation tests (CCTs) include prothrombin time/international normalised ratio (PT/INR), activated partial thromboplastin time (aPTT), platelet count, D-dimer and fibrinogen levels. While these parameters are important and widely used in the management of cardiovascular patients, CCTs are performed on platelet-poor plasma and are run in artificial states where the various blood elements are separated to allow for facility in performing assays, and these limitations should be recognised. CCTs cannot measure interactions between clotting factors, tissue factor and platelets. INR and PTT have the greatest utility in assessing patients being managed with anticoagulant therapy.
Several studies have questioned the usefulness and reliability of CCTs to assess coagulopathy and to guide haemostatic interventions, especially in the setting of perioperative bleeding.2,3 In certain patient categories (e.g. liver disease) where a complex balanced coagulopathy exists, these may be poor predictors of bleeding risk. Modest elevations of INR in the 1.3–1.8 range have been shown to be poor predictors of both bleeding and response to plasma therapy. While CCTs may identify patients at increased risk of bleeding due to thrombocytopenia, they do not provide data on qualitative platelet dysfunction, as may be seen, for example, with the use of antiplatelet agents or in uraemia. Bleeding time has grown out of favour for this purpose due to its operator dependence and lack of sensitivity.1,4 CCTs, thus, reflect a static evaluation of the coagulation cascade with clot formation as the endpoint rather than assessing the coagulation system as a whole. They have been shown to correlate poorly as predictors of clinical bleeding and transfusion requirements, fail to detect the effects of antiplatelet therapy or novel anticoagulation agents, do not describe platelet function or fibrinolysis, and lack accuracy in detecting deficiencies in coagulation factors.5,6
The limitations of CCTs, including the timeliness of reporting results, have led to the increased utilisation of viscoelastic haemostatic assays (VHAs), which depict a coagulation profile representative of the cell-based theory of haemostasis (i.e. initiation, amplification, propagation and termination through fibrinolysis).5 Its use in trauma patients (when compared with CCT) has allowed clinicians to better predict the need for massive transfusions, as well as mortality, and there has been a reported mortality benefit to thromboelastography (TEG)-directed haemostatic resuscitation in this population among patients requiring massive transfusions compared with those resuscitated with CCTs.7–10
A clinical benefit was also found in the haemostatic resuscitation of cirrhotic patients with non-variceal and variceal gastrointestinal bleeding, with a significant reduction in the number of transfused blood products when this was guided by TEG.11,12 TEG-guided blood product transfusion also demonstrated a lower rebleeding rate among cirrhotic patients with variceal bleed at 6 weeks.12 Its use in neurocritical care has allowed neurointensivists to elucidate different coagulopathy profiles among patients with intracerebral haemorrhage, subarachnoid haemorrhage and traumatic brain injury.13 More recently, it has been used with increasing frequency in the medical intensive care unit as a means of assessing the coagulopathy of coronavirus disease 2019 (COVID-19).14,15
Some studies have shown utility in predicting clinical course. For example, Mortus et al. showed elevated thromboses rates in patients with abnormal TEG parameters, and Wright et al. showed fibrinolysis shutdown, as demonstrated by elevated D-dimer, and complete failure of clot lysis at 30 minutes on TEG predicted venous thromboembolic events.15,16 In recent years, there has been a growing body of evidence supporting its use in critically ill cardiovascular patients. In this paper, we will review the fundamentals of VHA, its use and limitations, as well as identifying areas in need of future study in this patient population.
Overview of Viscoelastic Haemostatic Assay and its Interpretation
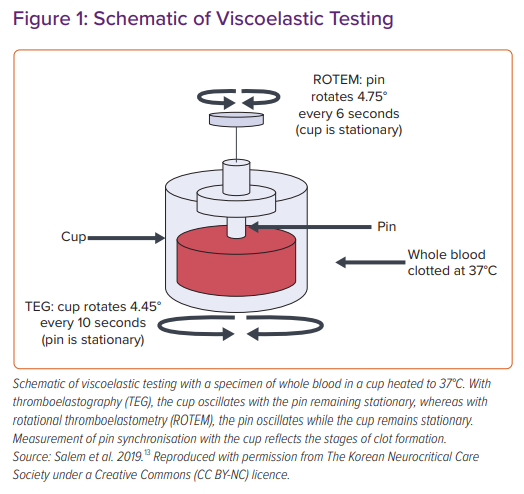
VHAs are performed through placing whole blood mixed with an activator (typically kaolin) and warmed to approximate body temperature (i.e. 37°C) in an oscillating cup with a suspended pin, which then transduces changes in viscosity determined by the tension in the pin. Changes in tension during clot formation and breakdown are plotted against time, and the resulting data provide a description of the coagulation and fibrinolytic profile of the sample tested (Figure 1). TEG is the VHA most commonly used in North America, with TEM/rotational thromboelastometry (TEM/ROTEM) presenting an alternative means of performing this test where the pin oscillates rather than the cup containing blood.
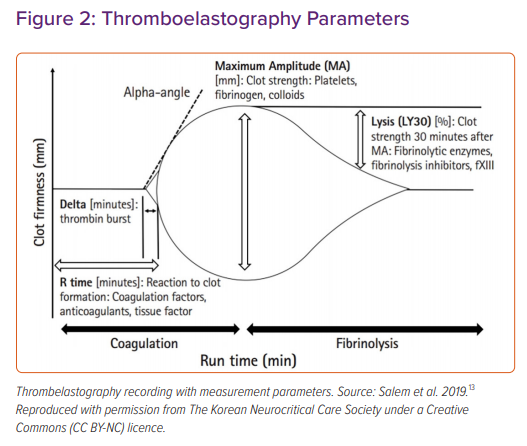
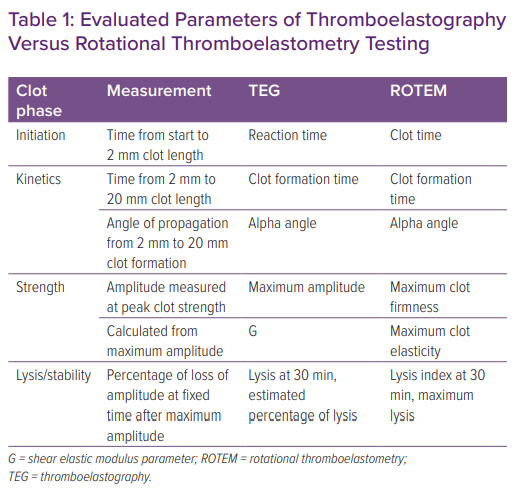
TEG and ROTEM measure different phases of the coagulation cascade. Measurements obtained from TEG include the time to initiate clot formation (reaction time; R), the rate of clot formation (kinetics; alpha angle), maximum clot strength (maximum amplitude; MA) and clot stability (fibrinolysis at 30 minutes; Figure 2 and Table 1).
ClotPro (Enicor, now acquired by Haemonetics Corporation, Boston, MA, US) is another VHA that uses thromboelastography with elastic motion. Similar to conventional thromboelastometry systems, the surfaces of the ClotPro cup and pin experience a relative movement, driven by an elastic element rotating the cup while the pin is stationary. The rotation of the cup is detected by a high-sensitivity electronic sensor. The ClotPro device comes with active tip technology, which eliminates manual reagent handling and improves standardisation. Both ROTEM and ClotPro include multiple panel assays that help us evaluate various aspects of coagulation cascade. For example, ROTEM assays include EXTEM, for evaluating the extrinsic pathway, INTEM, for the intrinsic pathway, FIBTEM, for evaluation of fibrinogen contribution to clot formation, and HEPTEM and APTEM for evaluation of heparin effect or thrombolysis reversal. HEPTEM and INTEM can be used together to demonstrate heparin-induced coagulopathy by evaluating clotting time on both the assays. FIBTEM and EXTEM used in conjunction can differentiate hypofibrinogenaemia and thrombocytopenia. EXTEM and APTEM when used in conjunction can diagnose fibrinolysis.
Quantra is a new VHA based on sonic estimation of elasticity via resonance sonorheometry technology.17 It is based on the principle that as the blood coagulates over time and increases its stiffness, the resonance frequency increases. These parameters are plotted over time. It evaluates the viscoelastic properties of whole blood by means of the following functional parameters: CT, CT with heparinase, clot stiffness, fibrinogen contribution to clot stiffness, platelet contribution to clot stiffness and CT ratio.
Sonoclot is a device that assesses viscoelastic properties for the blood using a sensitive electronic microviscometer that uses an oscillating suspended probe in the whole blood. It assesses haemostatic processes by assessing clot initiation by activated clotting time (ACT), fibrin propagation by clot rate and clot retraction using platelet function number. The platelet function number quantifies the quality of the clot retraction. Results will have values between 0 (no platelet function) and 5 (strong platelet function).
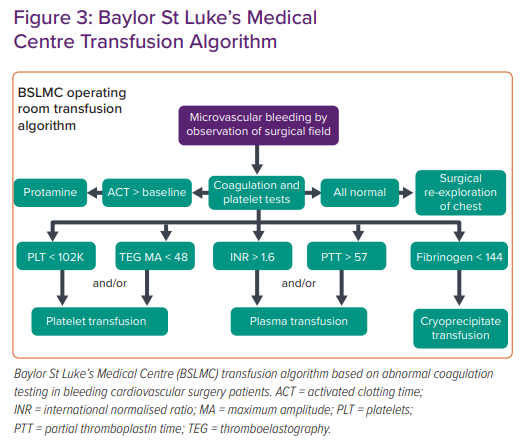
An example of a CCT- and TEG-guided transfusion algorithm implemented at the Baylor St Luke’s Medical Center (Houston, TX, US) cardiovascular intensive care unit is shown in Figure 3.
Limitations of Viscoelastic Haemostatic Assay
There are a few limitations to the use of TEG in cardiac patients, most notably its inability to reliably detect the presence of single antiplatelet therapy or warfarin.18,19 It may, however, detect the presence of combination antiplatelet therapy use. A variation of TEG, TEG with platelet mapping (Haemonetics Corporation) and ROTEM delta (Instrumentation Laboratory) with platelet assays, are specific VHAs that have been shown to correlate with platelet aggregometry, and are able to detect platelet dysfunction due to antiplatelet therapy and other coagulopathies.17,20 This test is discussed in further detail below in the subsection titled VHA in ACS.
It is also noteworthy that VHA testing is an in vitro assessment of coagulation, and thus does not factor in the role of vascular endothelium to coagulation; likely an important contributing factor in patients with cardiovascular disease. This test is also an inherently poor predictor of platelet adhesion and bleeding diathesis related to von Willebrand disease. Newer modifications of VHA (specifically ROTEM) with ristocetin have helped overcome this limitation; however, detection of mild-to-moderate von Willebrand disease is best done with other diagnostic systems.21 The coagulopathy induced by hypothermia during surgery, particularly cardiac surgery, is not detected by TEG, where the blood is warmed to a normal temperature at the time of testing; however, temperature adaptation can help overcome some of these challenges.22–24 The same limitations apply to CCT.
There is a need for multiple daily calibrations of these devices, performed by trained personnel and using standardised techniques, adequate maintenance and quality control, as well as standardisation of sample collection and testing to reduce interlaboratory variability. The newer generation of VHA, such as TEG-6, ROTEM sigma and Quantra, have cartridge-based automated test preparation without the need for manual pipetting and reagent mixing, which helps drive standardisation and reduces interpersonal and interlaboratory variability.
Viscoelastic Haemostatic Assay in Specific Cardiac Populations
Viscoelastic Haemostatic Assay in Cardiac Surgery
Cardiopulmonary bypass used in cardiac surgery causes several derangements in the haemostatic system.25 These derangements may lead to intraoperative and postoperative bleeding, mediastinal re-exploration, and transfusion of allogeneic blood products, all of which contribute to significant morbidity and mortality.26–28 Preprocedural factors, including dual antiplatelet therapy, oral anticoagulants that decrease thrombin production and hypofibrinogenaemia, also increase the risk of bleeding in cardiac surgery patients.29–31 Hence, timely diagnosis and treatment of any bleeding diathesis is imperative.32–34 Conventionally the decision to transfuse haemostatic blood products has been guided by clinical judgement or CCT. However, none of the standard laboratory coagulation tests were developed to predict bleeding risks or to guide coagulation management in surgical patients.35
A meta-analysis by Bolliger et al. evaluated 12 studies (two matched case–control, three retrospective cohort and seven randomised controlled trials) examining the role of TEG and ROTEM in the management of cardiac surgery patients.36 They found that TEG- or ROTEM-based transfusion triggers reduced the rates of blood component transfusions in cardiac surgery patients. Furthermore, there was a significant reduction in bleeding and surgical re-exploration after cardiac surgery.
A more contemporary meta-analysis by Meco et al. confirmed that VHA reduces blood component transfusions while also decreasing postoperative bleeding at 12 and 24 hours, and re-do sternotomies that were not due to surgical causes in cardiac surgery patients.37 Another meta-analysis by Dias et al. evaluated seven elective cardiac surgery randomised controlled trials. In the elective surgery meta-analysis, they showed reduced platelets, plasma transfusion, operating room length of stay, intensive care unit length of stay and bleeding rate.38 Hence, TEG- and ROTEM-guided transfusion protocols find themselves a 1C recommendation in the European Society of Anaesthesiology guidelines on the management of perioperative bleeding.39
Only a handful of TEG or ROTEM studies in cardiac surgery patients have reported mortality outcomes.40–42 Mortality was noted to be lower in the interventional arm in one of the studies, while two others did not show a mortality improvement with the use of VHA.42,43 Various factors may have influenced mortality outcomes in these studies, including patients’ baseline perioperative mortality risks (very low or very high), or analyses of only those patients that experienced bleeding. Furthermore, the primary aim of viscoelastic testing is to guide haemostatic interventions, which only indirectly affect mortality.
The use of VHA to predict bleeding in patients with symptomatic, severe aortic stenosis undergoing transcatheter aortic valve implantation was explored in a study of 54 consecutive patients by Rymuza et al. using TEG VHA testing.44 Samples drawn prior to the procedure were not predictive of bleeding complications. Receiver operating characteristic curve analysis of samples drawn at the end of the procedure showed significant specificity and sensitivity of bleeding complications; however, namely R, alpha angle and MA. After multivariate logistic regression analysis, MA was found to be an independent predictor of bleeding after transcatheter aortic valve implantation, both as a contiguous variable (OR 0.95 per 1 mm increment) and with a cut-off of ≤46.6 mm.
Viscoelastic Haemostatic Assay in Extracorporeal Membrane Oxygenation
Extracorporeal membrane oxygenation (ECMO) was first clinically employed in limited facilities in the 1970s, but in recent years the number of centres offering ECMO has increased dramatically.45,46 As the availability of ECMO has become widespread, the indications for its use have also expanded beyond cardiac failure and acute severe respiratory failure.47 Exposure of the patient’s blood to the various surfaces of extracorporeal circulation causes an inflammatory response, which triggers a systemic inflammatory response syndrome-like cascade, leading to an increased risk of both haemorrhagic and thrombotic complications.48 The rates of life-threatening haemorrhage and thrombosis in ECMO are reported to be between 10% and 33%, making anticoagulation and its monitoring a critical aspect of delivering ECMO support.49
Despite the widespread use of ECMO, there is significant variability among centres regarding anticoagulation monitoring, and currently no consensus exists for anticoagulation during ECMO. Unfractionated heparin is the anticoagulant of choice at most ECMO centres, and a survey of 121 international ECMO centres showed ACT to be the preferred (97%) method of anticoagulation monitoring.49 In addition to ACT, aPTT was used in 94% of patients at various intervals ranging from every 4–5 hours to >12 hours apart; routine or occasional antithrombin III (82%), anti-factor Xa (65%) and TEG (43%) testing during ECMO were also reported among surveyed centres.49 Among these various tests, TEG alone provides a comprehensive survey of haemostatic cascade, as discussed above.
Heparin’s impact on aPTT results is blunted by acute phase reactants; for example, alpha-2-macroglobulin and factor VIII. This may lead to an overdose of heparin when assessing anticoagulation with aPTT results only. Panigada et al. conducted a retrospective study of 32 patients treated with ECMO for severe respiratory failure to evaluate the prevalence of a TEG R >90 minutes (‘flat line’) reversible with heparinase during ECMO.50 They frequently observed a marked heparin effect on the TEG tracing despite an aPTT ratio (1.5–2.0) and ACT within the therapeutic anticoagulation range. These findings raise the concern that patients on ECMO may be excessively anticoagulated when utilising aPTT- and ACT-based protocols to guide heparin therapy.
In a follow-up study, Panigada et al. evaluated the safety and efficacy of TEG-driven heparin titration in ECMO patients.51 In a multicentre, randomised controlled trial, 42 patients with acute respiratory failure on veno-venous ECMO were randomised to either a TEG-based protocol (target 16–24 minutes of the R parameter, TEG group) or a standard of care aPTT-based protocol (target 1.5–2 of aPTT ratio, aPTT group) to guide heparin dosing. They found that heparin dosing was lower in the TEG group compared with the aPTT (p=0.03), while the number of haemorrhagic or thrombotic events and transfusions given were not statistically different between the two groups. However, there was a tendency for less bleeding from surgical sites and overall less bleeding in the TEG group.
Overall, TEG R-based heparin dosing for patients on ECMO appears to be safe, feasible and preferred over conventional aPTT-based dosing. In a small prospective observational study, Nair et al. studied ROTEM and platelet aggregometry, and suggested that ROTEM-guided coagulation management could avoid bleeding and possibly improve patient care.52 There is increasing interest in VHA testing in the management of patients on ECMO. However, there remains a paucity of high-quality evidence, thereby necessitating larger trials to determine the superiority of VHA testing compared with CCTs in this arena.
Viscoelastic Haemostatic Assay in Acute Coronary Syndrome
The most prominent event that defines acute coronary syndrome (ACS) is the formation of an intra-arterial thrombus, usually resulting from activation of platelets and fibrinogen at the ruptured plaque. A global haemostasis test, such as TEG, may show promise as a surrogate marker of the thrombus formation process and to aid in the diagnosis of ACS. Zhou et al. investigated its use for this purpose in a study of 142 patients with ACS, and found that the shear elastic modulus parameter (G), which is a computer-generated value reflecting complete strength of the clot and is calculated from the amplitude (A) with the formula: G = (5,000 × A) / (100 − A), is an independent diagnostic indicator of ACS (OR 2.6; 95% CI [2.035–3.322]) in this cohort of patients.53 The optimal cut-off value for the diagnosis of ACS was 10.55 dyne/cm2, while the sensitivity was 66.2% and the specificity was 92.4%.
Current guidelines recommend treatment with dual antiplatelet therapy for 6–12 months in all patients presenting with ACS or undergoing percutaneous coronary intervention and implantation of drug-eluting stents.54,55 The combination of aspirin and a P2Y12 inhibitor (such as clopidogrel, ticagrelor or prasugrel) is prescribed in these patients to prevent thrombotic events and adverse cardiovascular events.56 While the addition of P2Y12 inhibition to aspirin has significantly improved cardiovascular outcomes in patients presenting with ACS, platelet inhibitory responses to clopidogrel are subject to significant interindividual variability.57 Individual testing for clopidogrel hyporesponsiveness may be desirable, but is not routinely performed, because current gold standard tests for platelet reactivity, impedance aggregometry, light transmittance aggregometry and vasodilator-stimulated phosphoprotein phosphorylation assessment using flow cytometry, are time-consuming, require significant technical skill and are expensive.58,59
As discussed above, whole blood clot strength measured by TEG is not sensitive to platelet reactivity. Hence, the standard TEG has been modified to allow assessment of the contribution of P2Y12 receptor inhibitor by the addition of adenosine diphosphate (ADP), and the effects of aspirin by the addition of arachidonic acid (AA).60,61 This modification is referred to as platelet mapping, and is further modified to calculate the area under the curve at 15 minutes of the ADP trace.62 This modified TEG has been well validated to rapidly detect changes in platelet activity in response to loading doses of aspirin and clopidogrel.63–67 It has also been shown to correlate well with Accumetrics Verify-Now rapid platelet function analyser (r2=0.54, p<0.0001) and vasodilator-stimulated phosphoprotein phosphorylation (r2=0.26, p=0.001) to assess the response to clopidogrel in patients presenting with ACS.62
Reduced clopidogrel platelet inhibition and high residual platelet reactivity has been shown to lead to adverse outcomes in patients undergoing percutaneous coronary intervention for stable angina, unstable angina and ST segment elevation MI.68–71 AA- or ADP-induced platelet–fibrin clot strength (MAAA or MAADP) is indicative of the net residual platelet reactivity after treatment with aspirin or clopidogrel, respectively. A recently published post hoc analysis of a prospective, single-centre cohort study including 447 patients with ACS showed that the relative platelet inhibition rate (AA% or ADP%) independently predicted the risk of 6-month ischaemic events.72 Furthermore, high MAAA (HR 3.963; 95% CI [1.152–13.632]; p=0.029) and high MAADP (HR 5.185; 95% CI [2.228–12.062]; p<0.001) were independent predictors of ischaemic events, and an even higher risk rendered when they coexisted (HR 7.870; 95% CI [3.462–17.899]; p<0.001).
Viscoelastic Haemostatic Assay in Left Ventricular Assist Devices
Left ventricular assist devices (LVADs) improve survival and quality of life in end-stage heart failure patients who are refractory to medical therapy.73 This has led to a steady increase in their utilisation in recent years, with current annual implant rates exceeding 2,500/year.74 However, overall outcomes in these patients are significantly affected by two known complications of LVADs; namely, bleeding and pump thrombosis.75–77 The reported incidence of device thrombosis is around 8.4% at 3 months post-implantation, with an overall incidence of 12.3% at 24 months.75 The consequences of this complication can be severe, and include pump failure-induced cardiogenic shock, stroke and even death. The 6-month mortality associated with thrombotic complications in LVAD patients is high and reaches 48%.75
The prothrombotic milieu in LVAD patients is driven by a persistent high inflammatory state and endothelial activation leading to activation of clotting factors, as well as persistent platelet activation.78 Hence, pharmacological anticoagulation management after implant commonly includes a vitamin K antagonist in addition to a platelet inhibitor to decrease thrombotic and embolic risk. Current recommendations include warfarin dosed to an INR goal of 2.0–3.0 and an antiplatelet therapy, such as aspirin (81–325 mg daily).79 Adequate anticoagulation in LVAD patients is usually monitored through serial measurements of INR, and there exists limited data on the role of VHA in this patient population.
A single-centre retrospective analysis of 98 patients with durable mechanical circulatory support devices (31 Heartware LVADs, 25 HeartMate II [HM II] LVADs, 35 total artificial hearts [TAHs] and 7 biventricular assist devices) found the TEG-based coagulation index to be the single most statistically significant parameter used to optimally anticoagulate patients.80 The coagulation index is calculated using the reaction time, kinetics, alpha and MA values from a kaolin-activated TEG assay. In this study, a significantly higher coagulation index was observed among the patients that had thromboembolic events as compared with those who did not (mean for TAH3.12 versus 1.12, HM II 2.79 versus 1.74, Heartware 2.79 versus 1.70 and for biventricular assist device 2.79 versus 1.72). The authors of this study proposed that patients with HM II and Heartware devices should be maintained at a coagulation index value ≤1.5, whereas those with TAH devices should be maintained at a coagulation index ≤1.2 to minimise their risk of thromboembolic events. Furthermore, individualised INR goals should be set for patients based on what INR levels correspond to these coagulation index targets. These findings are consistent with another prior study involving patients with 99 SynCardia TAH.81
A more recent retrospective study by Xia et al. evaluated the role of TEG in predicting and defining pump thrombosis in HM II patients.82 A significant mean change in coagulation index of 0.71 (95% CI [0.1–1.32]; p=0.02) over a 24-month post-implantation follow-up period was noted in patients with suspected pump thrombosis compared with patients without. This change first became significant at 6 months. While the mean change in coagulation index significantly decreased over time in the group without pump thrombosis (−2.84; 95% CI [−5.21, −0.47]; p=0.02), it was not significantly different in the group with pump thrombosis (−1.72, 95% CI [−4.22, 0.78]; p=0.18). These findings make a case for routine TEG monitoring, specifically using mean changes in coagulation index, for evaluating pump thrombosis in HM II patients. Tarzia et al. reported a case of ROTEM-guided administration of recombinant activated factor VII for refractory bleeding after implantation of a biventricular assist device.83 Further prospective studies are required to validate these findings.
Viscoelastic Haemostatic Assay in Heart Transplants
Postoperative bleeding is one of the most common complications after cardiac surgery owing to the extracorporeal circulation, and contributes to significant morbidity and mortality. A single-centre observational prospective study of 49 cardiac transplant patients noted that the mean blood transfusion was 6.39 ± 5.33 units, fresh frozen plasma 4.9 ± 5.4 units and platelets 6.47 ± 9.61 units.84 Patients requiring ≥6 blood units were significantly more likely to require continuous renal replacement therapy (50% versus 12.5%; p=0.01) and had higher intensive care unit mortality (33.3% versus 4%; p=0.01).
Crabbe et al. reported a case of ROTEM-guided targeted haemostatic therapy in a heart transplant recipient who developed coagulopathy after therapeutic plasma exchange. Since VHAs offer better differential diagnosis of bleeding in the perioperative setting, they offer guidance for targeted haemostatic correction, as demonstrated by Crabbe et al.85
The key to prevention of major bleeding requiring massive transfusion in cardiac transplantation patients is adequate preoperative evaluation and analysis of bleeding or thrombotic tendencies and drugs that affect haemostasis. Although there is a paucity of literature on the utilisation of VHA testing in cardiac transplantation, it offers a promising strategy to guide individualised transfusion goals in this patient population.
Viscoelastic Haemostatic Assay in Direct Oral Anticoagulants
Direct oral anticoagulants (DOACs) are being used increasingly in patients with non-valvular AF for stroke prophylaxis, as well as venous thromboembolism prophylaxis and treatment. VHA have been used to assess the presence of DOACs in patients with acute bleeding events. Studies have shown prolongation of ROTEM clotting times and TEG Rs in the presence of DOACs.86–88 INTEM-CT is more prolonged compared with EXTEM clotting time, whereas maximum clot firmness is unaffected in the presence of DOACs.87,89 ROTEM and ClotPro have commercially available assays to detect direct thrombin inhibitors using their EcaTEM and Eca-Test assays, respectively. ClotPro has a commercially available Russell’s viper venom test assay to detect direct factor Xa inhibitors, while ROTEM modifications have been studied to evaluate activity of direct factor Xa inhibitors.90 There is also an investigational DOAC assay for the TEG 6s system that has been shown to have high sensitivity and specificity.90,91 VHA testing is promising in the setting of DOACs for their detection; however, there are no large clinical trials to demonstrate their utility in the role of assessing hard clinical endpoints.
Future Directions and Call for Further Areas of Study
There currently exists a paucity of data on the use of VHA in the setting of heart transplantation to guide transfusion strategies in this patient population. In the setting of ACS, further studies to validate the use of VHA as a diagnostic and prognostic aid are merited. As previously discussed, the use of TEG in patients with LVAD has revealed coagulation index to be an important parameter when determining the optimal degree of anticoagulation in this patient population. Further prospective studies examining the use of this parameter to guide anticoagulation against the incidence of thrombotic and bleeding events in patients with LVADs are required. Furthermore, the authors were unable to identify studies where VHA was utilised to assess the degree of platelet inhibition in this subset of patients.
While there exists a significant amount of data on the use of VHA with ECMO, there are currently three other types of temporary (or percutaneous) MCS devices; namely, the intra-aortic balloon pump, Impella devices and the TandemHeart.92 Anticoagulation with the use of an intra-aortic balloon pump is intended to reduce the risk of device thrombosis, thromboembolism or limb ischaemia. Current evidence on this is sparse, with some data suggesting that it may be safe to not anticoagulate when using intra-aortic balloon pump counter pulsation.93 The decision on whether or not to anticoagulate in this context should be tailored to the individual patient, on balance of the risks and benefits. VHA shows promise as a tool to guide such decisions; however, the authors were unable to identify any studies examining this. Impella and TandemHeart devices mandate anticoagulation to prevent device thrombosis.92,94 The prevalence of major bleeding complications among these patient populations is highly variable, with reports of up to 54% prevalence with Impella use and 59% with TandemHeart use.95 The authors could not identify any studies that incorporate the use of VHA in the management of anticoagulation in these settings, which may guide clinicians in identifying those that are at a heightened risk for complications.
Conclusion
CCTs have long been used to guide the anticoagulation status of critically ill and perioperative cardiovascular patients; however, they present many limitations. VHA overcomes many of these limitations, and although it has been in use for >60 years, its use in this patient population remains relatively nascent. It has been shown to reduce the need for blood component transfusions, as well as postoperative bleeding and mediastinal re-exploration when used to guide transfusions in the surgical setting. In the ECMO population, its use has been associated with lower doses of unfractionated heparin use when compared with CCT-guided protocols, with no associated increase in the incidence of thromboembolic events and a trend towards fewer bleeding events. Due to its properties in defining the patient’s overall haemostatic profile, VHA shows promise in many other applications within cardiovascular critical care. There is a need for further studies exploring the use of VHA in this arena.