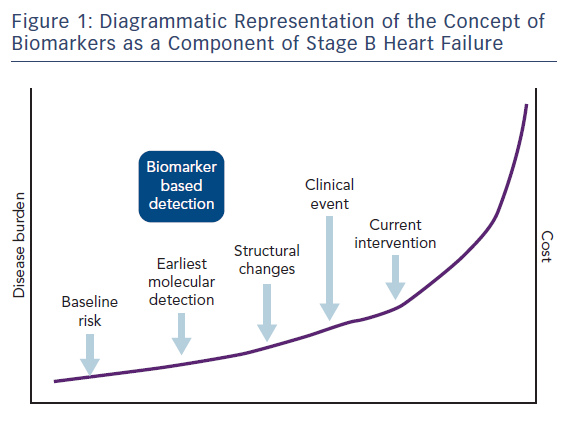
Increasingly biomarkers are of interest in cardiovascular disease (CVD) for risk stratification. In particular, natriuretic peptides (NPs), which were originally used for the diagnosis of heart failure, are now finding a role in identifying those most at risk of heart failure and other cardiovascular (CV) disorders. Their ability to be measured rapidly through blood tests makes their widespread use more practical. They may also aid in the detection of disease at an earlier stage before structural and functional changes become apparent on imaging (see Figure 1).
Natriuretic Peptides
Several data sets1–3 indicate that NPs are effective in refining risk prediction for CVD and add predictive power to conventional risk factors. Conventional risk indicators (e.g. lipids or hypertension) reflect potential for CV damage, whereas early elevations of NP are an endogenous response to often preclinical CV damage, which allows time for intervention. In addition to standard signals for NP release, such as volume overload, other work4 has demonstrated this peptide responds to fibro-inflammation, a fundamental pathophysiological signal present from the outset of many CVDs and indeed comorbidities such as cognitive impairment and ischaemia.5,6
Increases in plasma brain natriuretic peptide (BNP) or N-terminal prohormone BNP (NT-proBNP) concentration have diagnostic and prognostic implications in selected populations, as demonstrated initially in heart failure, and subsequently in early-stage and asymptomatic CVD. Recent reports have suggested that NP provides prognostic information for a wide variety of CVDs beyond that obtained from routine risk factors. A recent study7 showed NT-proBNP is predictive of future CHD and stroke in individuals without known CVD at the time of measurement. The individual participant data meta-analysis included 40 prospective cohorts comprising over 95,000 individuals. It also suggested the risk prediction with NT-proBNP is greater in older compared with younger individuals.7 The estimation of personal CVD risk in older individuals is difficult using current population-based models, due to the higher incidence of CVD in this group. Levels of B-type NP and N-terminal pro-atrial NP strongly predicted the risk of heart failure, with an increase in the adjusted risk of 77 % and 94 %, respectively, per one standard deviation increment in log peptide values.
Studies on Natriuretic Peptide-based Screening and Prevention
Two trials have tested the approach of using NPs as part of a strategy to identify those at highest risk of CV events and targeting treatment to these groups in order to prevent heart failure and other CV disorders. Both these studies – St. Vincent’s Screening to Prevent Heart Failure (STOP-HF) and NT-proBNP Selected PreventiOn of cardiac eveNts in a populaTion of dIabetic patients without A history of Cardiac disease (PONTIAC) – had favourable results.
The STOP-HF trial was a pragmatic randomised controlled trial involving one specialist centre and 39 general practices with 1,374 participants. Those included were asymptomatic individuals >40 years old with a history of one or more of the following: hypertension, hyperlipidaemia, obesity, vascular disease (coronary artery disease, cerebrovascular disease and peripheral vascular disease), diabetes mellitus, arrhythmia requiring therapy or moderate to severe valvular disease.
Participants were randomised to a control group (receiving routine general practitioners [GP] management and specialist care as required) or BNP-driven collaborative care between the GP and specialist CV centre. In the intervention group, BNP results were made available to GPs, with protocol-driven referral to the specialist CV service, of all patients with a value of ≥50 pg/ml. Those with BNP values <50 pg/ml received the same care as provided in the control group but with disclosure of BNP values to patients and GPs. Participants with a BNP level of ≥50 pg/ml underwent echocardiography and review by a cardiologist at the study centre, who decided on further investigation and management. The focus of the specialist intervention for those with elevated BNP was multidimensional and included optimal risk factor management and complete investigation and treatment of abnormalities defined on examination or on echocardiography. In addition, all patients received further coaching by a specialist nurse who emphasised individual risk status and the importance of adherence to medication and healthy lifestyle behaviours.
A total of 263 patients (41.6 %) in the intervention group had at least one BNP reading of ≥50 pg/ml. The intervention group underwent more CV investigations (control: 496 per 1,000 patient-years versus intervention: 850 per 1,000 patient-years; incidence rate ratio 1.71; 95 % CI [1.61–1.83]; p<0.001) and received more renin–angiotensin– aldosterone system (RAAS)-based therapy at follow up (control: 49.6 %; intervention: 56.5 %; p=0.01). The primary endpoint of left ventricle (LV) dysfunction with or without heart failure was met in 59 (8.7 %) of 677 in the control group and 37 (5.3 %) of 697 in the intervention group (OR 0.55; 95 % CI [0.37–0.82]; p=0.003). Asymptomatic LV dysfunction was found in 45 (6.6 %) of 677 control-group patients and 30 (4.3 %) of 697 intervention-group patients (OR 0.57; 95 % CI [0.37–0.88]; p=0.01). Heart failure occurred in 14 (2.1 %) of 677 control-group patients and 7 (1.0 %) of 697 intervention-group patients (OR 0.48; 95 % CI [0.20–1.20]; p=0.12). The incidence rates of emergency hospitalisation for major CV events were 40.4 per 1,000 patient-years in the control group versus 22.3 per 1,000 patient-years in the intervention group (incidence rate ratio 0.60; 95 % CI [0.45–0.81]; p=0.002). Interestingly there were no statistically significant differences in risk factor control between the control and intervention group at the end of the study. Similar effects have been seen in other positive studies on CVD.8 A subsequent analysis of the cost-effectiveness of this approach was undertaken. The cost per quality-adjusted life year gain was €1,104 and the intervention has an 88 % probability of being cost-effective at a willingness to pay threshold of €30,000.9
In the STOP-HF study it was noted the benefits observed in the intervention group were likely multifactorial. They include facilitating targeted therapy changes, in particular increased used of angiotensin receptor blockers and increased use of diagnostic tests. Although blood pressure reduction in both groups was similar, the targeted use of RAAS-modifying therapy may have contributed to the reduction in endpoints through mechanisms other than blood pressure reduction. Patient adherence to therapy and lifestyle advice may have been encouraged by communicating risk status to patients.
In the PONTIAC trial, 300 patients with type 2 diabetes, increased level of NT-proBNP (>125 pg/ml) but free from CVD were randomised to either standard treatment at diabetes care units or an ‘intensified’ strategy in which patients were additionally treated at a cardiac outpatient clinic for the up-titration of RAAS inhibitors and beta-blockers. Cardiac disease-based exclusion criteria were one or more of the following: history of cardiac disease; signs of cardiac disease on the ECG such as AF; ST-T-wave abnormalities or a bundle branch block; abnormal echocardiography (with the exception of diastolic dysfunction) defined as low ejection fraction; wall motion abnormalities; significant valve dysfunction or other significant alteration.
The primary endpoint of hospitalisation/death due to cardiac disease at 2 years was significantly reduced with use of the intensified strategy (hazard ratio 0.35; 95 % CI [0.13–0.98]; p=0.044). After 12 months there was a significant difference between the control and intensified groups in both the number of patients treated with renin–angiotensin system (RAS) antagonists and beta-blockers and in the dosage reached (p<0.0001 for all). RAS antagonists were up-titrated to 100 % of the recommended dosage in 79 % of cases in the intensified group compared with 42 % in the control group (p<0.0001). Beta-blockers were up-titrated to 100 % of the recommended dosage in 51 % of cases in the intensified group and in only 10 % of cases in the control group (p<0.0001). A combination of 100 % of the RAS antagonist and 100 % of the beta-blocker recommended dosage was achieved in 46 % of cases in the intensified group and in 5 % of cases in the control group (p<0.0001).
Similar to STOP-HF, blood pressure was significantly and similarly reduced in both groups after 12 months (p=0.003 control group; p=0.002 intensified group). Heart rate was reduced only in the intensified group (p=0.004) and there was a trend towards this also in the STOP-HF study (p=0.06). Similar to the STOP-HF study there was no significant changes in NP concentrations.
Inclusion in the Guidelines
Both the STOP-HF and PONTIAC randomised clinical trials are included in the 2014 Canadian Cardiovascular Society Heart Failure Management Guidelines10 as a recommendation for the use of NPs in at-risk individuals. The guideline suggests for individuals with risk factors for the development of heart failure, NP levels be used to implement strategies to prevent heart failure. In the guideline it is recommended an increased level of NP of BNP >100 pg/ml and NT-proBNP >300 pg/ml be used to avoid overscreening. However, this does not appear to be based on any analysis and is not supported by the cost-effectiveness analysis carried out by the STOP-HF team.9
In more recent guidelines from the US11 it is recommended for patients at risk of developing heart failure, NP biomarker-based screening followed by team-based care, including a CV specialist optimising guideline-directed medical therapy, can be useful to prevent the development of left ventricular dysfunction (systolic or diastolic) or new-onset heart failure. It highlights the need for further studies to determine cost-effectiveness and risk of such screening, as well as its impact on quality of life and mortality rate.
Future Directions
The data above highlights the emerging role of NP intervention in international health systems and the need to evaluate the intervention further by comparing to current interventions for heart failure prevention.
There is increasing interest in the concept of personalised medicine, whereby conditions with common pathophysiologies are targeted together, and also using biomarkers to identify patients who will most benefit from certain interventions. Approaches such as those used in STOP-HF12 and PONTIAC13 involve personalised medicine using NPs to identify those patients most at risk of CV events from a broad group of conditions, such as hypertension, diabetes, ischaemic heart disease, AF and cerebrovascular disease, and targeting care to the group defined as high risk from within this cohort. In particular, PONTIAC also intensified use of RAAS and beta-blocker therapies in a predetermined fashion. STOP-HF did not have a predefined strategy but did have intensification of RAAS therapy in the intervention group noted at study end. By using molecular markers of disease these approaches identify abnormalities earlier allowing for detection and intervention at an early stage. The ARNI in Asymptomatic Patients With Elevated Natriuretic Peptide and Elevated Left Atrial Volume Index eLEvation (PARABLE) study14 is investigating the use of the angiotensin receptor-neprilysin inhibitor sacubitril/valsartan in those with hypertension and/or diabetes and an elevated NP level. This is based on the concept of using a biomolecular signal to initiate a particular therapy targeted at that biomolecular pathway and represents a study based on the personalised medicine concept. Its primary outcome is left atrial volume index measured by cardiac MRI.
Common Pathophysiological Processes and Personalised Medicine
CVD causes 1.9 million deaths (40 % of all deaths) annually in the EU, and is estimated to cost the EU economy almost €196 billion a year.15 In the setting of the continuing burgeoning rise in both risk factors and CVD, the continuation of directing resources in a non-discriminatory manner poses a major threat to the sustainability of healthcare systems and heightens inequality.
Ongoing difficulty in preventing cardiometabolic diseases and improving outcomes may in part be explained by the uniform direction of resources to a population containing predominantly lower-risk patients. Currently, prevalent approaches to heart failure prevention devote the same level of resources to all individuals in a broad population (typically, a population exhibiting risk factors such as obesity, smoking, or conditions such as diabetes or hypertension). NP-based screening and targeted prevention are a possible approach to this issue.
New Research
The emergence of this strategy of NP screening and targeted intervention requires further study of a number of factors, such as: determining optimal levels of NPs for risk stratification, determining the role of other biomarkers (troponin, ST2 and galectin-3 or multiplex panels in refining risk prediction), the development of clinical prediction rules integrating clinical features and biomarkers to improve risk prediction, the evaluation of alternative systems of care using this approach (increasing the role of generalist physicians or using alternative systems to obtain specialist advice, such as web conferencing),16 determining the optimal use of medications (RAAS inhibitors and beta-blockers) and refining the need for cardiac imaging in this cohort.
Conclusion
The use of NPs to identify individuals who would most benefit from heart failure prevention strategies holds great promise. By targeting care to those most at risk it provides a strategy for sustainable heart failure prevention models. This approach is now part of international guidelines and further studies to refine this model of care are ongoing.







